 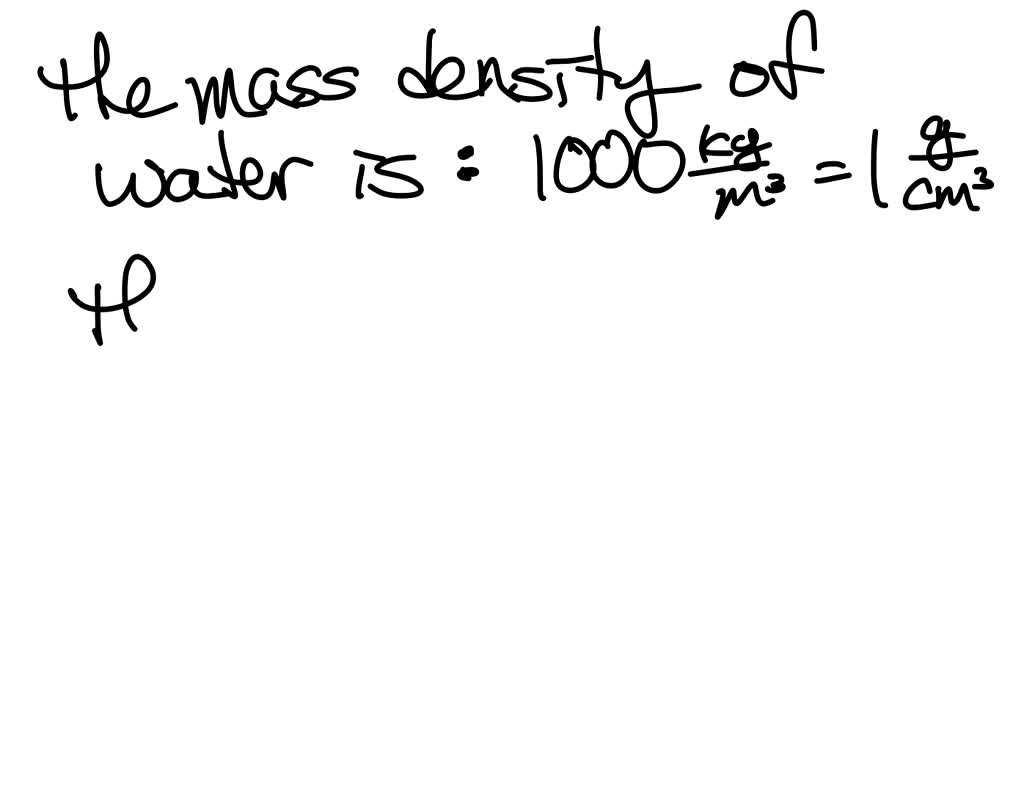 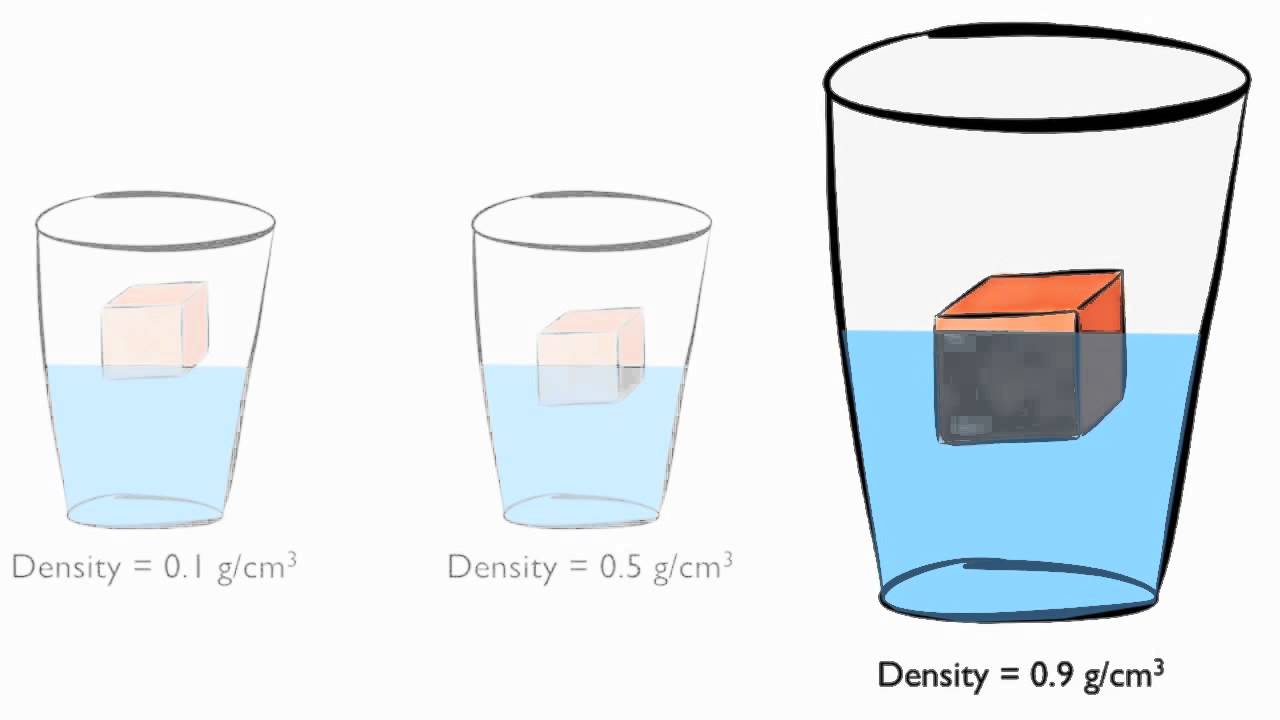 
(Left) Crystal structure of ice, with water molecules held in a regular 3D structure by hydrogen bonds. Their difference is the buoyant force \(F_B\). Water is an anomaly (that is, a weird standout) in its lower density as a solid. This pressure and associated upward force on the bottom of the cylinder are greater than the downward force on the top of the cylinder. Remind students to wash their hands after completing the activity.Īll common household or classroom materials can be saved or disposed of in the usual manner.\): Pressure due to the weight of a fluid increases with depth since \(P = h\rho g\). Make sure you and your students wear properly fitting safety glasses or goggles. The activity sheet will serve as the Evaluate component of the 5-E lesson plan. Ice is less dense than liquid water, so it floats. The maximum density of water occurs around 4 degrees Celsius. Note that water can be supercooled and remain a liquid well below its normal freezing point. A standard table lists the values for the density of liquid water. Students also learn that if an object is more dense than water it will sink when placed in water, and if it is less dense than water it will float.ĭownload the student activity sheet and distribute one per student when specified in the activity. The density of water is about 1 gram per cubic centimetre (62 lb/cu ft): this relationship was originally used to define the gram. The density of pure water actually is somewhat less than 1 g/cm 3.The density of water is most given as 1 g/cm3, but below is the density of water with different units. Students are also introduced to the idea that whether a substance sinks or floats in water is a characteristic property of that substance and does not depend on the amount of the substance. Density is the mass per unit volume of a substance.Students are introduced to the concept of density and that density has to do with how heavy something is relative to its size.Assessment does not include density or distinguishing mass and weight.”Īlthough the standard does not call for density to be used as a characteristic property to identify a substance, a basic introduction to density is included here as an optional element of a learning progression leading up to a middle school understanding of density. There are two main factors that make ocean water more or less. density) that are independent of sample size. Note: The NGSS Structure and Properties of Matter for 5 th grade, Performance Expectation 5-PS1-3 states “density is not intended as an identifiable property. Sea water has characteristic properties (e.g. NGSS 5-PS1-3: Make observations and measurements to identify materials based on their properties.If your students have already learned the meaning of volume and mass, you can easily use those terms to define density as Density = mass/volume and then use those terms in the lesson. We are also using “heavy”, “light”, and “weight” instead of “mass”. Note: We are purposely using the terms “size” and “amount” instead of “volume” in discussions of density. Density is a characteristic property of a substance and doesn’t depend on the amount of substance.If an object is more dense than water it will sink when placed in water, and if it is less dense than water it will float.As you can see in the chart, water only has an exact density of 1 g/cm 3 at 39.2F or 4.0C. The maximum density of water is 0.9998395 g/ml at 39.2° F (4. Water Density at Different Temperatures Below is a chart that shows the density of water (in grams/cm 3) at different temperatures, ranging from below water's freezing point (-22F/-30C) to its boiling point (212F/100C). We know water density can be expressed in different units, such as lb/ft 3 or g/ml, or g/cm 3, but the actual density of water is not 1 g/ml, a bit less than 1. Density is a measure of how heavy something is compared to its size. Well, you must remember a fact about water density.Students will also be able to explain that density is a characteristic property of a substance. Students will be able to explain that the density of a substance has to do with how heavy it is compared to the size of the object. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
